Tiny Dancers
Samuel Stupp, PhD, has discovered how to control the collective motion of more than 100,000 “dancing” molecules within a nanofiber — a revolutionary strategy that has the potential to reverse spinal cord injuries and more.
by Gina Bazer
Regenerative medicine is a flourishing area at Northwestern, and Samuel Stupp, PhD, director of Northwestern’s Simpson Querrey Institute for BioNanotechnology (SQI), is often leading the charge. He has been for more than two decades — he founded SQI in 2000 as the Institute for Bioengineering and Nanoscience in Advanced Medicine.

Stupp’s laboratory engineers molecules that self-assemble into nanofibers, which mimic cell structures and biological signaling. The technology can be used for everything from cartilage repair to regenerative therapies for organ transplantation to potential treatments for inflammatory bowel disease. The possibilities are virtually limitless, so it is no surprise that scientists across disciplines have partnered with Stupp on novel solutions to the vexing challenges in their fields.
Stupp’s latest breakthrough is a promising therapeutic approach that can reverse paralysis and repair tissue after severe spinal cord injuries. For this, he leveraged technology — an injection therapy made up of self-assembling peptide amphiphiles, or PAs — that he pioneered two decades ago. In a study published in Science this past fall, Stupp and his team administered a single injection of the PA formula into the tissues surrounding the spinal cords of paralyzed mice, spurring targeted cell signaling by molecules with intense motion that promoted rapid healing. Just four weeks later, the animals regained the ability to walk.
“Currently, there are no therapeutics that trigger spinal cord regeneration, and I wanted to tackle this problem,” says Stupp, who is also the Board of Trustees Professor of Materials Science and Engineering, Chemistry, Medicine, and Biomedical Engineering. “We are going straight to the FDA to start the process of getting this new therapy approved for use in human patients.”
“Our fundamental discovery about controlling the motion of molecular assemblies to enhance cell signaling could be applied universally across biomedical targets.”
Samuel Stupp, PhD
‘Dancing molecules’ hit moving targets
The secret behind Stupp’s new breakthrough therapeutic is tuning the motion of molecules, so they can find and properly engage constantly moving cellular receptors. Injected as a liquid, the therapy immediately gels into a complex network of nanofibers that mimic the extracellular matrix of the spinal cord. By matching the matrix’s structure, mimicking the motion of biological molecules, and incorporating signals for receptors, the synthetic materials can communicate with cells.
“Receptors in neurons and other cells constantly move around,” Stupp says. “The key innovation in our research, which has never been done before, is to control the collective motion of more than 100,000 molecules within our nanofibers. By making the molecules move, ‘dance’ or even leap temporarily out of these structures, known as supramolecular polymers, they are able to connect more effectively with receptors.”
Stupp and his team found that fine-tuning the molecules’ motion within the nanofiber network to make them more agile resulted in greater therapeutic efficacy in paralyzed mice. They also confirmed that formulations of their therapy with enhanced molecular motion performed better during tests with human cells, indicating increased bioactivity and cellular signaling.
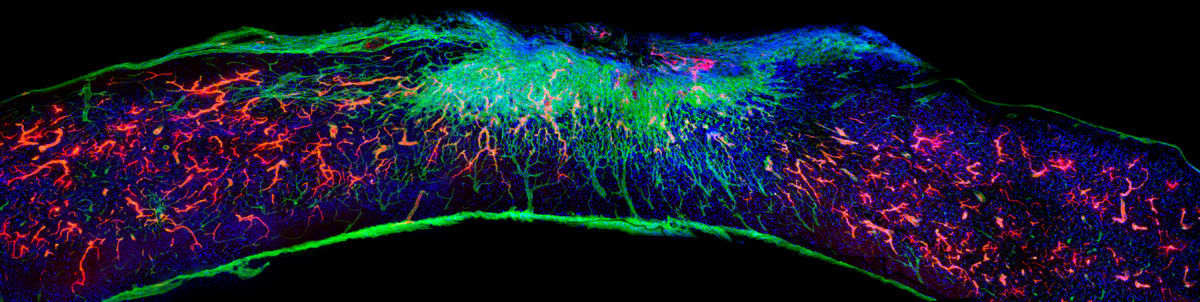
“Given that cells themselves and their receptors are in constant motion, you can imagine that molecules moving more rapidly would encounter these receptors more often,” Stupp says. “If the molecules are sluggish and not as ‘social,’ they may never come into contact with the cells.”
Once connected to the receptors, the moving molecules trigger at least two cascading signals, both of which are critical to spinal cord repair. One signal prompts the long tails of neurons in the spinal cord, called axons, to regenerate. Like electrical cables, axons send signals between the brain and the rest of the body. Severing or damaging axons can result in the loss of feeling in the body or even paralysis.
Repairing axons, on the other hand, increases communication between the body and brain. The second signal helps neurons survive after injury because it causes other cell types to proliferate, promoting the regrowth of lost blood vessels that feed neurons and other critical cells for tissue repair.
Universal application
While the new therapy could be used to prevent paralysis after major trauma (automobile accidents, falls, sports accidents and gunshot wounds) as well as from diseases, Stupp believes the underlying discovery — that “supramolecular motion” is a key factor in bioactivity — can be applied to other therapies and targets.
“The central nervous system tissues we have successfully regenerated in the injured spinal cord are similar to those in the brain affected by stroke and neurodegenerative diseases, such as ALS, Parkinson’s disease, and Alzheimer’s disease,” Stupp says. “Beyond that, our fundamental discovery about controlling the motion of molecular assemblies to enhance cell signaling could be applied universally across biomedical targets.”
Other Northwestern authors of the Science study include Evangelos Kiskinis, PhD, assistant professor in the Ken and Ruth Davee Department of Neurology and of Neuroscience; research technician Feng Chen; postdoctoral researchers Ivan Sasselli, PhD; Alberto Ortega, PhD; and Zois Syrgiannis, PhD; former graduate students Alexandra Kolberg-Edelbrock, PhD, and Stacey Chin, PhD; and graduate student Ruomeng Qiu. Peter Mirau, PhD, of the Air Force Research Laboratories; and Steven Weigand of Argonne National Laboratory also are co-authors.
To learn more, listen to our Breakthroughs podcast with Stupp.
Connecting Investigators
SQI and its Center for Regenerative Nanomedicine connect investigators across disciplines to foster translational research partnerships at Northwestern, other U.S. institutions, and around the globe with the goal of improving human health, well-being, and quality of life. A current multi-institution collaboration, involving investigators from Northwestern, the University of Wisconsin, Mayo Clinic, and the Steadman Philippon Research Institute in Vail, Colorado, is focused on accelerating cartilage repair following arthroscopic hip and knee procedures. This work was spearheaded by Northwestern trustee Michael Shannon.
32
faculty members are members of SQI
472
PhD students, medical students, postdocs, and fellows trained at the institute
5
start-ups have been launched as a result of SQI research

Advancing Organ Tissue Repair
The Feinberg Cardiovascular and Renal Research Institute (FCVRRI) is another breeding ground for regenerative medicine study at the medical school. A recent grant through NUGoKidney is funding a partnership between Stupp and Lorenzo Gallon, MD, professor of Medicine in the Division of Nephrology and Hypertension and of Surgery in the Division of Organ Transplantation. The laboratories are working together to develop peptide amphiphile-coated regulatory T-cells to use in a mouse model of kidney ischemia-reperfusion injury, with the long-term goal of using this strategy to prevent ischemia-reperfusion injury, and thus improve outcomes after organ transplantation.



FCVRRI scientists are also making innovative inroads by studying the regenerative properties of lymphatics. Guillermo Oliver, PhD, director of the institute’s Center for Vascular and Developmental Biology and the Thomas D. Spies Professor of Lymphatic Metabolism, is exploring how lymphatics can help to restore heart function after cardiac injury (he was senior author of a study published in Nature in 2020 finding that lymphatics promote cardiac growth and repair in mice). Meanwhile, Michael Donnan, ’13 MD, ’16, ’18 GME, an instructor in the Division of Nephrology and Hypertension, is studying the active role lymphatics play in the response to kidney injury, exploring the possibility of targeting kidney lymphatics for regeneration and/or repair of the organ.

Pioneering Regenerative Engineering
Biomaterials play a critical role in the field of regenerative medicine, especially in the replacement of damaged tissues — an area of study that is also growing at Northwestern, particularly in the laboratory of Guillermo Ameer, ScD, the Daniel Hale Williams Professor of Biomedical Engineering at the McCormick School of Engineering and professor of Surgery in the Division of Vascular Surgery at Feinberg.
Ameer’s laboratory is dedicated to the development of biomaterials and nanotechnology for regenerative medicine, specifically tissue engineering, medical devices, drug delivery, and cell delivery applications. A medical product based on a citrate-based biomaterial technology called CITREGEN™, pioneered in Ameer’s laboratory, will soon be widely available for use in reconstruction surgeries that involve attaching soft tissue grafts to bone.
This past fall, Ameer was elected to the National Academy of Medicine (NAM). Membership in the academy is one of the highest honors given to a scientist in the United States.
Amanda Morris contributed to this story.