Keeping the Beat
by EMILY AYSHFORD
Northwestern’s new Center for Arrhythmia Research will develop novel ways to treat irregular heartbeats.
The opening of the new Northwestern University Center for Arrhythmia Research this year underscores just how far treatment and detection of irregular heartbeats has come since the electrocardiogram (EKG) was developed in the 19th century. At the time, it the was one of the first instances of technology providing physicians with objective data about what exactly was going on within our internal organs.
In 1909, the system was used to diagnose atrial fibrillation as the cause of an irregular heartbeat. Atrial fibrillation — commonly known as AFib — happens when the heart’s upper chamber beats out of sync with its lower chamber. This arrythmia can come and go, or it may be persistent. In both cases, the resulting low blood flow can lead to clots, stroke, and heart failure.
Over the past 100 years, physicians and investigators have developed therapies to treat AFib, often through blood thinners (which can reduce clots), or through interventions like ablation, which destroys tissue in the heart that may be causing this irregularity. But as our lifespans grow longer, AFib has become more common. More than six million Americans are currently living with the condition — a number that is expected to rise to more than 12 million by 2030 due to an aging population.
The risk factors from AFib vary greatly from person to person, which makes personalized care imperative. That’s why, thanks to a generous $10 million gift from several donors, Feinberg is opening the new center. Through the center, teams of interdisciplinary clinicians and scientists will work together to discover both the underlying molecular causes of arrhythmias and new standards of care for treatment.
And though the electrocardiogram is still used in clinics today, physicians and investigators now have many more technologies at their disposal — such as implantable monitors and Apple Watches — to bring a new level of detection and care to their patients.
“Northwestern is uniquely positioned to be a worldwide leader in this field,” says Rod Passman, MD, professor of Medicine in the Division of Cardiology and of Preventive Medicine, and a cardiac electrophysiologist at Northwestern Memorial Hospital, who will direct the new center. “My goal is to change the way we manage these diseases to improve the lifespan and life experiences of our patients.”

We want to be able to use the technology available to us to better understand stroke prevention and to perhaps spare patients from being on blood thinners for the rest of their lives.
Rod Passman, MD
Determining Who is at Risk For Stroke
Passman has always been interested in using the latest techniques and technology to provide the best surveillance and care for his patients since he joined the field as a professed lifelong learner.
“I don’t think anyone says as a child, ‘I want to be a cardiac electrophysiologist when I grow up,’” says Passman, who is also the Jules J. Reingold Professor of Electrophysiology. “But I wanted to pick the area in medicine that made the least sense to me, that would be the most challenging, so it would hold my interest.”
Nearly 30 years later, Passman is highly regarded in the field and is a leader in cardiac monitoring and stroke prevention. He helped pioneer the use of implantable cardiac monitors for AFib in stroke patients, and he recently published a study in the journal Circulation that used data from device-detected AFib to measure the risk of stroke. By linking two databases — the electronic records of more than 11 million patients and a database of 300,000 patients with cardiovascular implantable electronic devices that could detect AFib — they found that not all cases of AFib lead to stroke.
By measuring the stroke risk of 21,000 patients using the CHA2DS2-VASc score — a clinical score that takes into account risk factors such as age and cardiovascular history — they found that only AFib of an increased duration was associated with a higher risk of stroke, even among those with other similar stroke risk factors.
“If you have a low amount of atrial fibrillation, and not a lot of other risk factors, your risk of stroke is very low,” Passman says. That’s important because most patients with AFib are treated the same way — a lifetime of blood-thinning drugs, which can be expensive and have unwanted side effects.
“The way we treat atrial fibrillation today is based on historical measures, and we don’t individualize care very much,” Passman says. “We want to be able to use the technology available to us to better understand stroke prevention and to perhaps spare patients from being on blood thinners for the rest of their lives.”
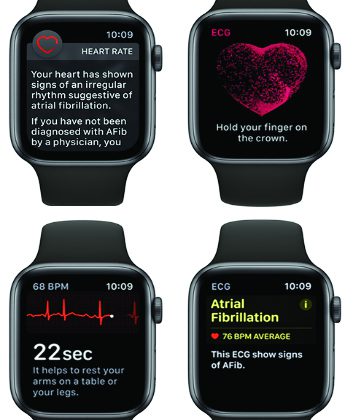
A Watch That Can Alert You to AFib
Passman showed in a recent study that implantable cardiac monitors could reduce patients’ time on blood thinners by 95 percent. But those monitors are not scalable to the entire population of AFib patients. They cost thousands of dollars and often only send data to clinicians once a day, in the middle of the night.
Instead of developing new technology, electrophysiologists are looking toward consumer products, like Apple Watches, which now have the ability to check heart rhythm. That means the watch could alert wearers when they are in AFib and then remind them to take their blood thinner. (Recent advances mean blood thinners can now thin blood within a couple hours of administration.)
Passman is in the final phases of evaluations for a large study that would test how well this system could work. Patients enrolled in the study would each receive an Apple Watch and would use the watch’s pulse checker to track their AFib.
“It’s a wearable AFib monitor that’s built into a consumer grade electronic device for less than 1 percent of the cost of an implantable device,” he says.
Understanding AFib at All Levels
Passman and his colleagues are also searching for the underlying factors and mechanisms that cause AFib. In 2018, the American Heart Association awarded Feinberg a $3.7 million grant as one of six centers in a national network dedicated to researching and understanding the causes of atrial fibrillation.
Understanding this disease extends to both epidemiological studies and basic science research. As part of the grant, Philip Greenland, MD, the Harry W. Dingman Professor of Cardiology, director of the Center for Population Health Sciences, and a professor of Preventive Medicine in the Division of Epidemiology, is studying cardiac structure and blood flow across populations to determine AFib prevalence and stroke risk.
At the same time, Rishi Arora, MD, professor of Medicine and director of Experimental Cardiac Electrophysiology in the Division of Cardiology, is studying the underlying molecular mechanisms of AFib and developing new gene-based therapies for the disorder. By using novel gene therapy approaches to target the fundamental molecular mechanisms underlying AFib, such as inflammation and increased “firing” of the heart’s intrinsic nervous system, he and his laboratory team hope to prevent progression of AFib and potentially even cure the disease. “We want to create viable therapies to be able to target these fundamental mechanisms very soon,” Arora says.

You have to understand what’s normal to best understand how abnormal a flow might be.
Michael Markl, PhD
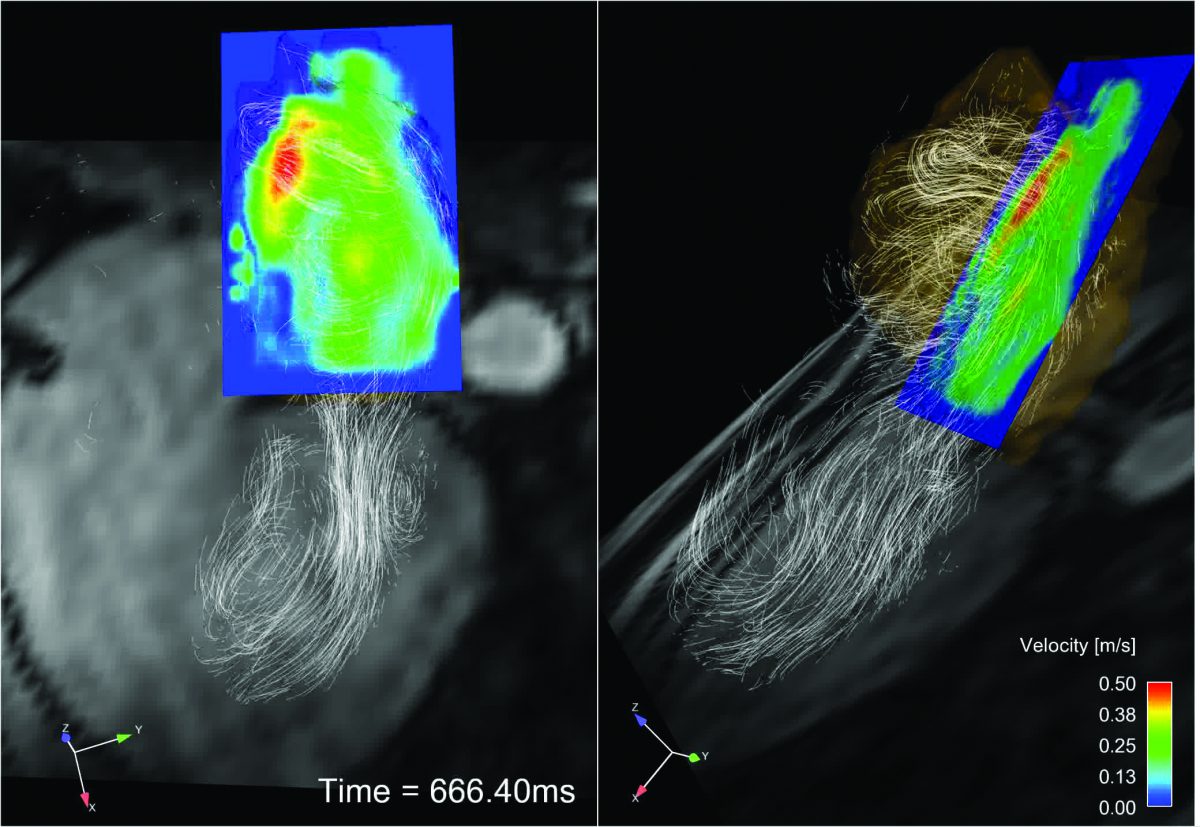
The investigators also collaborate with Michael Markl, PhD, vice chair for Research in the Department of Radiology, the Lester B. and Frances T. Knight Professor of Cardiac Imaging, and a professor of Radiology. A physicist by training, Markl has developed a new imaging technique that can measure the 3D blood flow in the heart without any radiation or contrast agents. Called 4D Flow MRI, the technique can measure blood flow through thousands of images. Markl and his collaborators are using their imaging tool to create a more patient-centric risk metric for stroke risk. Clinicians like Passman use the technique, which takes 5-10 minutes as part of a regular cardiac MRI, to understand how patient risk changes if they’ve had ablation or other therapies. “Sometimes, even if you cure the arrythmia, you don’t restore normal blood flow,” Markl said. “We want to know if this group is still at continued risk for stroke.”
Markl is also examining flow across a wide range of patients. “You have to understand what’s normal to best understand how abnormal a flow might be,” he says.
New Center Will Be Catalyst for Research
It’s collaborations like these that have fueled the founding of the new center. The center will bring together faculty from across disciplines — cardiology, radiology, engineering, and computer science — to better understand and treat arrhythmia disorders.
Conditions like these require a collaborative approach, Arora says. “At Northwestern, we are able to look at the disease from the molecular level all the way to the patient,” he says. “We need a formal center of this type to enable investigators to more closely communicate with each other and to foster productive, cross-disciplinary collaborations among investigators at Feinberg and other schools at Northwestern, like the McCormick School of Engineering.”
The center hopes to attract the best students and investigators through fellowship programs, and will provide catalyst funds for early-stage research at the university. It will also help Northwestern Medicine stay at the leading-edge of treatment. “We treat a lot of patients, and we want to tell patients that we are not only a great place for clinical treatment but we also are the place where cutting-edge research is done, where new treatments are going to come from,” Arora says.

We want to tell patients that we are not only a great place for clinical treatment but we also are the place where cutting-edge research is done, where new treatments are going to come from.
Rishi Arora, MD